 This dark space extends through the Tube at further low pressure appearing Fuzzy. 3 – Cathode Ray Tube Experiment for Thomson’s Atomic ModelĪt low pressure of about 1 millimetre of mercury, Cathode Rays appeared slightly divergent and dark near the cathode. 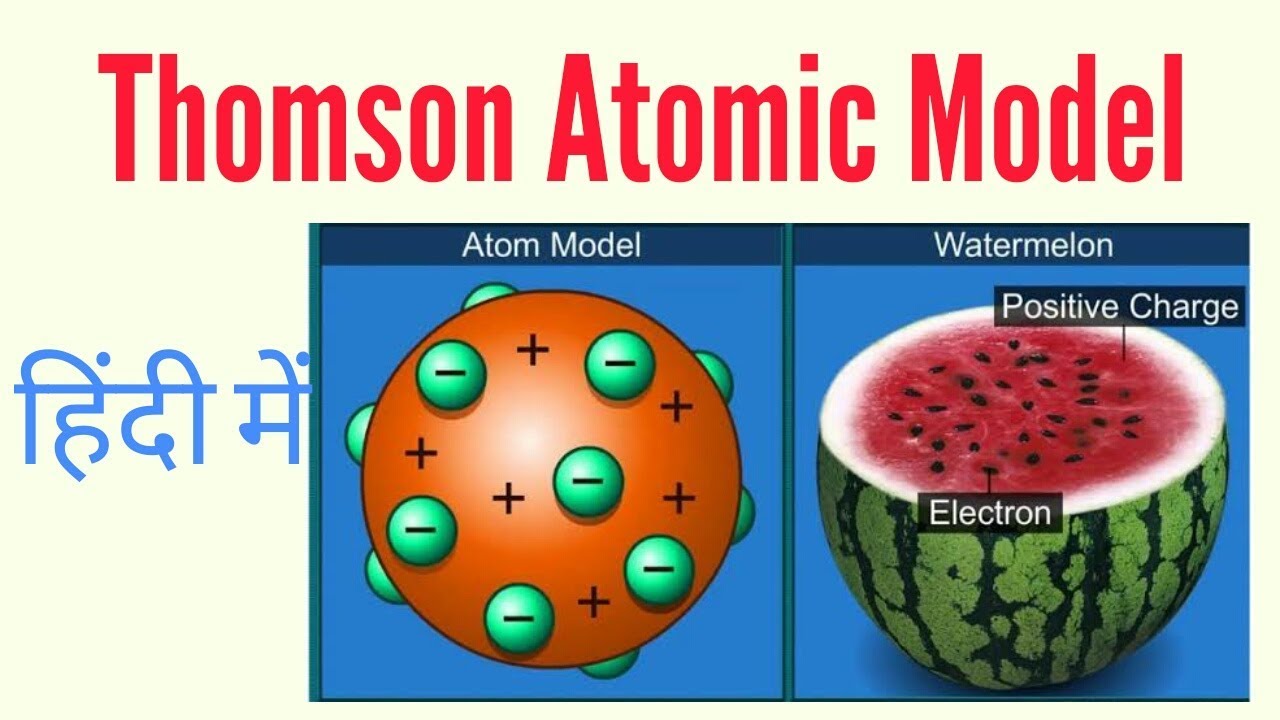
He also identified that by changing and adjusting the strengths of Electric and Magnetic field, the Cathode Rays did not deflect.įig. He found that the beam could be deflected by electric field while turning off the Magnetic field. Electrons from Cathode on the left side of the Tube travels through the slit (Anode), and comes in contact with the Phosphorescent screen at the right end of the Tube. 3 shows the Schematic representation of the same. The apparatus built by him was called the Cathode Ray Tube which is popularly known today as ‘ Electron Gun’.įig. It was an attempt to conclude that Cathode Rays carried negative charge. Thomson conducted series of experiments to examine the nature of Cathode Rays. Since the positive and negative charges are equal in magnitude, an Atom is electrically neutral.An Atom consists of a positively charged sphere with electrons encased in it. 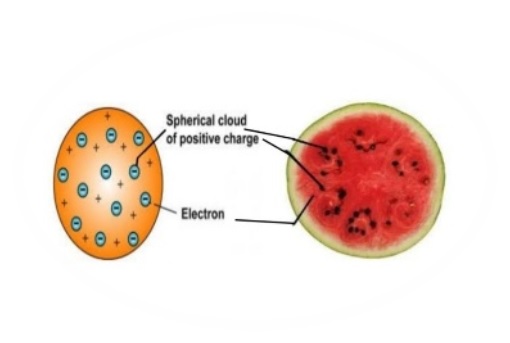
2 – (a) Schematic Representation of Thomson’s Atomic Model (b) Atomic Model Compared to Plum Pudding Postulates of Thomson’s Atomic Model Hence the name Plum Pudding Model was attributed to Thomson’s Atomic Model.įig. This scientific model was compared to Plum Pudding, where the distribution of Electrons was considered as ‘Plums’ (Raisins) in a positively charged Space which was referred to as Pudding. He also assumed that these Negative ions or Electrons moved in an orbit in a sphere which had equal positive charges. Thomson’s Atomic Model states that an Atom is electrically neutral and is made up of ‘Corpuscles’ which act as Negative ions. 1 – Introduction to Thomson’s Atomic Model He called the ‘Electrons’ as “ Corpuscles”.įig. 
His theory states that an Atom is made up of thousands of Electrons and an Electron is 2000 times lighter than Proton. He conducted his experiment using Cathode Ray Tube (CRT). Thomson’ was a great physicist who has the credit of discovering ‘Electrons’ and was awarded Nobel Prize for his discovery. Thomson’s Atomic Model (also called as ‘ Plum Pudding Model‘) explains the inner structure of the atom theoretically. Thomson’s Model, How does Plum Pudding Model Work, applications and limitations. This post will discuss what is Thomson’s Atomic Model, postulates of J.J. Thomson’s Atomic Model also called as ‘ Plum Pudding Model’ was the most accepted Atomic Model during the year 1904-1910, which emphasized on the inner structure of the Atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
